Water treatment equipment is used to clean and purify water by removing harmful substances such as pollutants, impurities, and microorganisms. The goal is to make the water safe and suitable for different uses by meeting specific quality standards.
In general, water treatment equipment includes the following parts:
Pretreatment systems, quartz sand filters, activated carbon filters, ion exchangers, membrane systems, ozone, and UV disinfection systems.
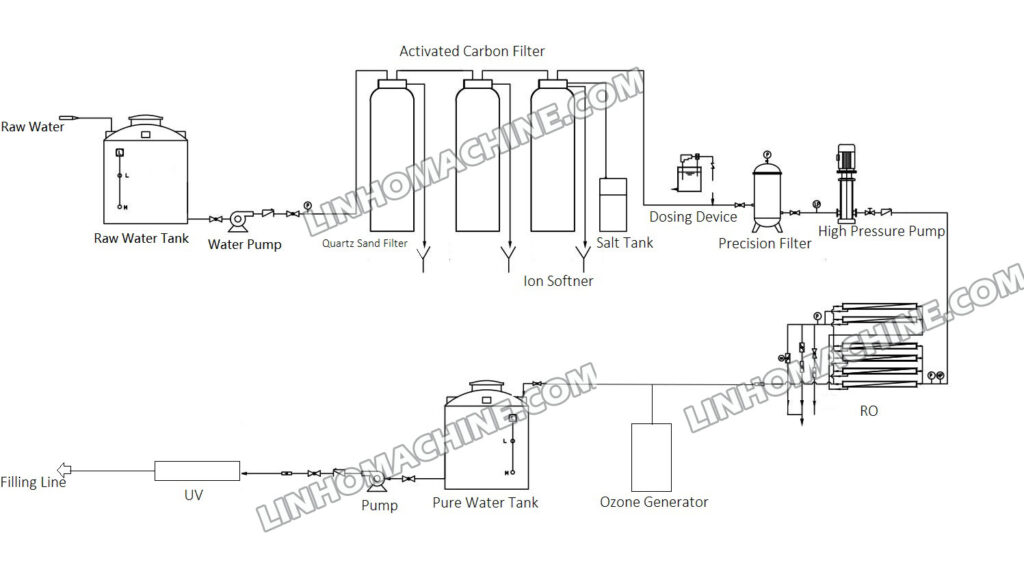
1. Pretreatment system: The pretreatment system is responsible for removing suspended particles, sediment, organic matter, and other impurities in water by using physical or chemical methods. The equipment used for this purpose includes grilles, sedimentation tanks, coagulators, and filters.
2. Quartz sand filter: The quartz sand filter performs depth and fine filtration. When water is the primary raw material for beverage production, using quartz sand filters is advisable to maintain water quality purity and stability.
3. Activated carbon filter: The activated carbon filter eliminates odors, organic pollutants, and assorted colors in water. It is made of high porosity and surface area activated carbon that efficiently absorbs organic matter, chlorine, and chloramines in water. This results in an improvement in the taste and safety of the water.
4. Ion exchanger: The ion exchanger removes hardness ions such as calcium, magnesium, and iron from water to prevent scale buildup and pipe blockages. There are two types of ion exchangers: anion exchangers and cation exchangers, which use different exchange resins based on water quality requirements.
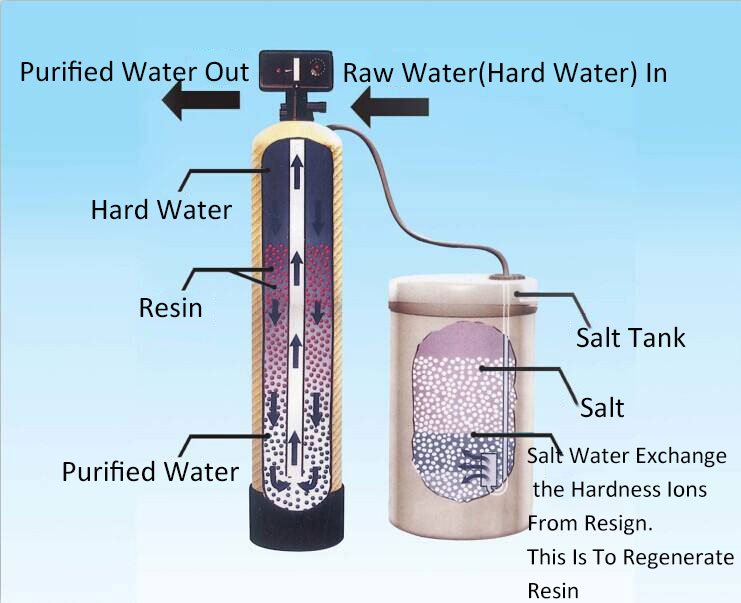
Ion exchangers typically comprise a resin column filled with ion exchange resin. When water flows through the resin column, the ions in the water exchange with the ions in the resin, resulting in the removal or conversion of ions in the water.
The specific operation process of the ion exchanger is as follows:
Pretreatment: Pretreat the water to remove impurities, including suspended solids.
Softening treatment: During the ion exchange process, water flows through the resin column, where the sodium ions in the resin column exchange with the calcium and magnesium ions in the water. This results in the removal of hardness ions from the water, achieving a softening treatment.
Remove other ions: Remove other ions in the water, such as chloride ions, iron ions, etc. as needed.
Regeneration: Ion exchange resins lose their effectiveness over time and require regeneration to restore their performance. During regeneration, a specific concentration of brine passes through the resin column, replacing the ions in the resin and restoring its adsorption capabilities.
Over time, the ion exchange resin barrel loses its exchange capacity, leading to poor water quality and the need to replace the resin. The replacement time of the resin depends on several factors, including the water’s hardness, purpose, and the type of resin used. Regular water hardness testing is essential, and the resin should be replaced based on actual conditions. Typically, the resin barrel should be replaced every 3-5 years.
Ion exchange is an effective method for removing impurities and ions from water, resulting in pure water that is suitable for various industries, including pharmaceuticals and industrial production.
5. Ultrafiltration, nanofiltration, and reverse osmosis membrane systems are advanced and mature technologies used to remove microorganisms, organic matter, dissolved solids, and other impurities from water, resulting in pure or high-purity water. These membrane systems are widely used in the field of water treatment due to their effectiveness and versatility.
Ultrafiltration is a separation technology that physically isolates impurities, macromolecules, colloids, bacteria, and other harmful substances from water using ultrafiltration membranes. The ultrafiltration membrane acts as a screen, allowing small molecular substances like dissolved inorganic salts and organic substances to pass through while retaining larger, harmful substances.

Reverse osmosis is a membrane separation technology that uses semi-permeable membranes to selectively allow water and solutes to pass through from a high concentration solution to a low concentration solution via natural osmosis. Applied pressure can also be used to reverse the direction of water flow, allowing water to pass from a low-concentration solution to a high-concentration solution in a process called reverse osmosis.
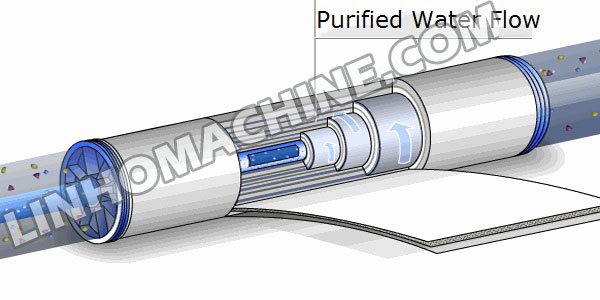
Reverse osmosis plants use a semi-permeable membrane to pretreat and filter water. This membrane allows only water molecules to pass through, while solutes are blocked. When high pressure is applied, water molecules are filtered out through the membrane, leaving behind solutes and impurities, resulting in the production of pure water.
Nanofiltration is a separation and filtration technology that utilizes membranes with nanoscale pore sizes. These membranes usually have pore sizes between 1 and 100 nanometers, smaller than those used in ultrafiltration. With its ability to filter tiny impurities, ions, and macromolecular organic substances, nanofiltration is ideal for water, beverage, food, and pharmaceutical treatment.

Nanofiltration differs from ultrafiltration and reverse osmosis in terms of separation mechanisms and application scenarios. The key difference is the size of the membrane pores and the separation mechanism. Nanofiltration has smaller pores than ultrafiltration and can effectively remove small molecules and particles. Compared to reverse osmosis, nanofiltration is a more economical and straightforward method that doesn’t require as much energy.
Ultrafiltration membranes have a pore size range of 0.001-0.1 microns and can effectively separate substances with a molecular weight greater than 1000 Daltons, such as bacteria, viruses, proteins, and colloids. The separation mechanism of ultrafiltration is primarily based on the size, shape, and charge characteristics of the substances that pass through the membrane.
Nanofiltration uses membranes with pore sizes less than 0.001 microns to filter and remove substances with molecular weights greater than 200 Daltons, such as glucose, amino acids, and electrolytes. The membrane works based on the size, shape, and charge characteristics of the substances, separating them from the solvent or smaller molecules.
Reverse osmosis relies on semi-permeable membranes with a pore size typically below 0.0001 microns to separate nearly all dissolved substances in water, including salts, heavy metals, bacteria, viruses, and more. The separation mechanism is based on the selective permeability of water and solutes through the membrane.
Ultrafiltration and nanofiltration are commonly used in the food industry for processes such as concentration, decolorization, desalting, and deacidification of beverages, dairy products, and fruit juices. On the other hand, reverse osmosis has a broader range of applications, including water production, wastewater treatment, seawater desalination, and the electronics industry. To summarize, ultrafiltration and nanofiltration have larger pore sizes compared to reverse osmosis, resulting in less efficient separation but lower equipment and energy costs. On the other hand, reverse osmosis has smaller pore sizes and better separation efficiency, but equipment and energy costs are higher. Choosing between these technologies should be based on the specific needs and cost-effectiveness of the application.


